A high-throughput approach to detect pathogenic bacteria:
Identifying pathogenic bacteria (bacteria that cause diseases in humans) in natural samples has important consequences for human health. For example, 80% of foodborne illness comes from "unknown origin" --- there are either too few pathogenic bacteria in the sample to detect, or there does not exist a specific test for the pathogen at fault. To address these concerns, we focus on high-throughput methods that require no prior knowledge of the bacteria to assess its pathogenicity. Our current approach relies on droplet microfluidics: we co-encapsulate human tissue with the bacteria of interest, forming a functional assay capable of screening 106 bacteria per day.
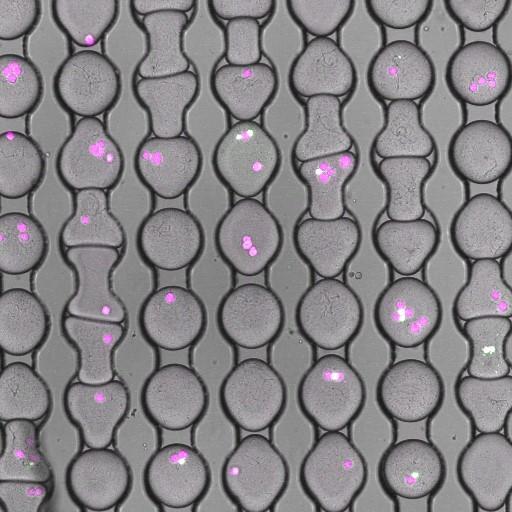
Figure 1: 200 micron diameter droplets immobilized on an array for time-lapse imaging. The droplets contain mammalian cells and pathogenic bacteria. The mammalian cells are stained with a live fluorescent marker (magenta) at time zero and then encapsulated in the droplets with the bacteria and a dead stain (green). The “white” cells in the image are dead; the color is a combination of the magenta live dye and the green dead dye.
Better organoids through homogeneity:
Organoids are 3-dimensional masses of cells that better reproduce in vivo function than tradition 2-dimensional cell culture. They currently show promise as sources of therapeutic cells, as better testing platforms for new drugs, and as platforms to better understand organ development in general. However, one problem with current protocols to produce organoids is the large variability in the mature organoids. This variability makes it difficult to do quantitative studies with organoids and consequently, realize their full potential. We are attempting to solve this problem using droplet microfluidics. The basic idea is to specify the initial conditions for each individual organoid such that they all identical at the start of their development. The hope is that a homogeneous starting condition leads to a more homogeneous mature organoid population. To get a homogeneous starting population, we randomly encapsulate starting cells into aqueous droplets and then sort for the droplets with the desired number and composition of starting cells. Currently, we are working on both the microfluids and the biology for this initial step.

Figure 2: Cluster of human embryonic stem cells encapsulated into droplets. These clusters form 1-6 hours after encapsulation and will eventually develop into an embryoid body, an organoid model of early human development.
Contact: Perry Ellis pellis@g.harvard.edu
